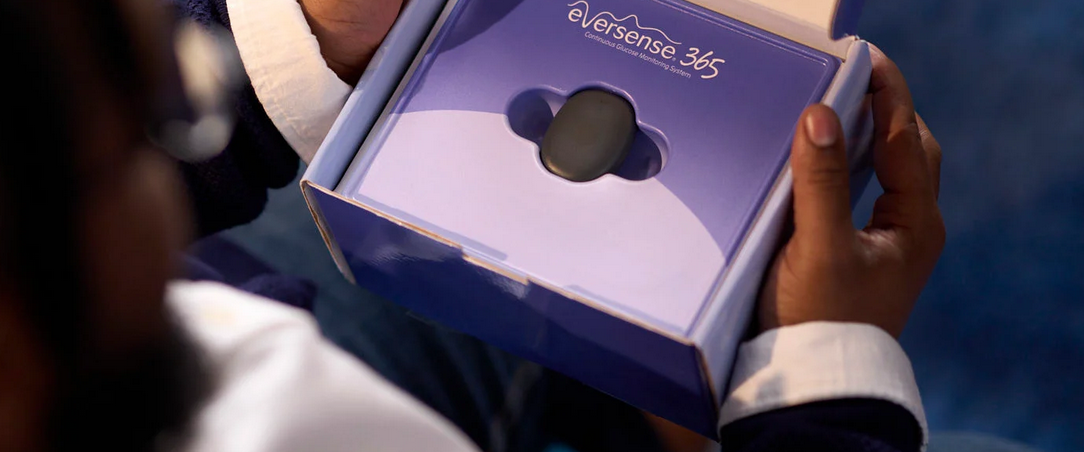
Eversense 365 - A 365-day implantable CGM has just launched in Europe
The global rollout of the Eversense 365 system in the European market is gaining momentum. It is the first continuous glucose monitor that only needs to be implanted once every 365 days. After a successful debut in the USA (October 2024), it’s time for the Old Continent. The launch in Sweden is just the beginning – Germany, Spain, and Italy are next in line.
In this post, you’ll find all the key information about Eversense 365 – from specifications and the implantation process to how it compares with Dexcom G7 and Libre 3.
What is Eversense 365 and how does it differ from traditional CGMs?
Eversense 365 is an implantable CGM system (Continuous Glucose Monitor) by Senseonics. The main difference compared to standard sensors (like Libre 3 or Dexcom G7) lies in its design and wear duration:
Standard CGMs – you stick the sensor onto your skin (arm, abdomen) and replace it every 7–14 days. Each new sensor means another insertion and another cost.
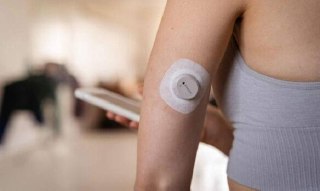
Eversense 365 – a small sensor (about 1.8 cm long) is placed by a doctor under the skin of the upper arm during a short outpatient procedure. The sensor works for a full 365 days, after which the doctor removes it and (optionally) implants a new one.
Key features of Eversense 365
Feature | Eversense 365 | Libre 3 / Dexcom G7 (for comparison) |
|---|---|---|
Sensor lifespan | 365 days | 14 days (Libre 3) / 10 days (G7) |
Location | Subcutaneous (implant) | Attached to the skin |
Calibration | Required once daily | None (factory calibrated) |
Reading frequency | Every 5 minutes | Every 1–5 minutes |
External transmitter | Yes (magnetic, worn on the skin above the sensor) | No (built into the sensor) |
Alerts | Yes, via app | Yes, via app |
The external transmitter is currently the only “minor drawback” – it needs to be charged daily (about 15 minutes) and worn on the skin. But remember – the sensor itself can operate without it for some time, storing data.
Availability in Europe – where and when?
According to Senseonics’ announcement from April 24, 2026, the rollout looks like this:
Sweden – already available to first patients (April 2026)
Germany, Spain, Italy – planned launches in the coming weeks
More countries – as part of Senseonics’ growth strategy, which aims to expand the target market by over 30 million patients with type 1 and type 2 diabetes (18+)
At the time of writing, there is no official information about reimbursement in individual countries. In the USA, the retail price of Eversense 365 (sensor + transmitter + implantation service) is about $1999 per year. In Europe, we can expect similar pricing, but much will depend on negotiations with public payers.
Will Eversense 365 be available in Poland?
So far, there are no specific announcements regarding Poland. Considering that Germany is one of the first markets, and Poland often follows Western European trends, availability in 2027 is possible. I’m monitoring the topic and will share updates when the first signals from a distributor appear.
What does implantation and daily use look like?
This process differs significantly from the “stick and forget” approach of traditional CGMs. Here’s what you need to know:
Implantation procedure – performed by a doctor (usually a diabetologist or surgeon) in an outpatient setting. The skin on the upper arm is locally anesthetized, then a small incision (about 5 mm) is made and the sensor is placed. The whole process takes about 10–15 minutes. The sensor is invisible to the naked eye.
First hours – for 24 hours after the procedure, you must not get the arm wet. Afterwards, for about 2–3 days, there may be slight swelling or bruising – this is normal.
Transmitter – once the wound heals, you place a flat transmitter on the skin (magnetically). It communicates with the sensor and sends data to the mobile app. The transmitter is charged daily for about 15 minutes (e.g., during a shower).
Calibration – the system requires one daily fingerstick calibration (glucometer reading). For some, this is a downside; for others, an opportunity for verification. Senseonics claims this calibration improves long-term accuracy.
Replacement – after 365 days, the doctor removes the old sensor (a similar small procedure) and can immediately implant a new one.
Eversense 365 vs Dexcom G7 vs Libre 3 - a quick comparison
Since I document apps and devices for glucose monitoring on this blog, I know many of you compare available solutions. Eversense 365 is not a typical “competitor” to G7 or Libre 3, as it addresses slightly different needs.
Who is Eversense 365 better for?
People with frequent sensor detachment (sweating, adhesive allergies, active lifestyle)
Users who don’t like changing sensors every 7–14 days
Patients with skin issues at typical CGM application sites
Those seeking discretion – the sensor is completely invisible.
Who is better off with a traditional CGM (Libre 3 / Dexcom G7)?
Those who don’t want daily fingerstick calibration
Those who don’t want to wear an additional removable transmitter
Those who prefer a disposable sensor without doctor visits for replacement
When a lower upfront cost matters (although the annual cost of Eversense may be comparable or lower than 26 Libre 3 sensors).
An important note about the future: Senseonics is already working on next-generation systems – Gemini and Freedom – which will eliminate the need for an external transmitter. When these hit the market, Eversense will become even more competitive.
Does Eversense 365 work with insulin pumps and apps?
Senseonics announced that in the USA, Eversense works with the twiist pump (a hybrid closed-loop system). In Europe, there is currently no information about integration with popular pumps (e.g., MiniMed 780G, Tandem Mobi, Ypsomed). However, the Eversense app (iOS and Android) displays all glucose data, trends, alerts, and reports. You can also export data to Tidepool, Glooko, or a doctor’s web portal.
If you use glucose monitoring apps like xDrip+, Diabox, or Juggluco – the community is already working on integration with Eversense 365. At the time of the European launch, only the Eversense app and selected partners are officially supported.
Summary - is Eversense 365 worth considering?
Pros:
One procedure for the entire year (360+ days of peace of mind).
No visible sensor on the skin between replacements.
Less waste (one small implant vs. 26 Libre 3 sensors per year).
Stable readings – the sensor won’t detach during workouts or bathing.
Cons:
Requires daily fingerstick calibration.
Wearing and charging the external transmitter daily.
Doctor visits for implantation and removal (a drawback for some).
Higher barrier to entry (cost, availability, required medical approval).
My opinion (based on documenting apps and CGMs): Eversense 365 is a technological breakthrough, but not for everyone. If you have skin issues, frequently lose or damage sensors, or are simply tired of weekly replacements – it’s worth considering. If you value minimal invasiveness and don’t mind changing sensors every 14 days – stick with Libre 2/3 or G6/G7.
Sources and further information
Official Senseonics announcement (via BioSpace) – European launch of Eversense 365
Article Diabetech.info – Eversense 365 Implantable CGM Officially Launches in Europe
Have you already used Eversense 365 or are you considering implantation? Let me know in the comments – I’ll gladly share additional information or help you find a center in your country.