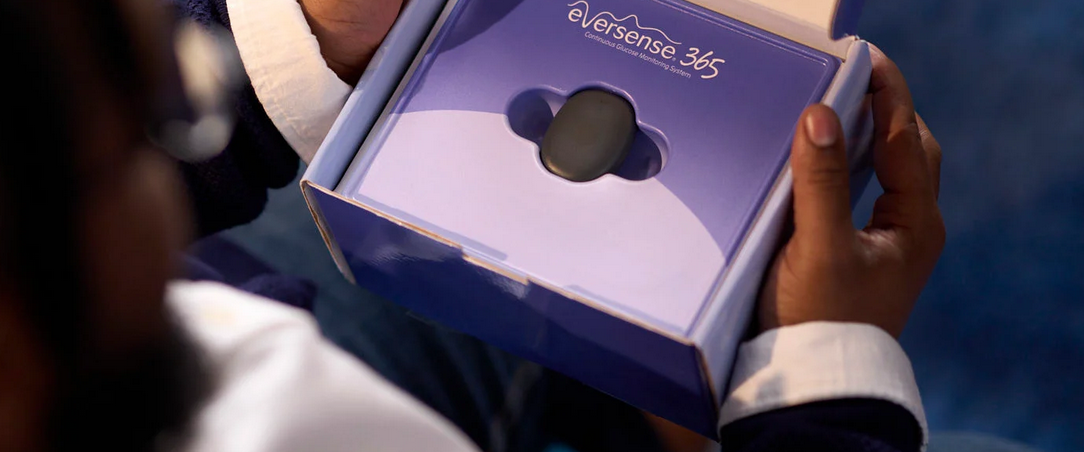
What is Simplera Sync and how does it differ from its predecessor?
Over the past few years, users of the MiniMed 780G pump have mainly used the Guardian 4 system, which required a separate transmitter (the so-called “shell”) placed on the sensor and regularly charged. Simplera Sync completely changes this model: the sensor, transmitter, and applicator are combined into a single, integrated, disposable device.
What does this mean in practice? Fewer components to manage, less charging, less risk that something will run out of power in the middle of the night. The sensor is disposable and works for up to 7 days, and all the equipment needed for insertion is already in the box.
Key technical features – what you should know
Before we move on to user experience, a few hard facts:
Compatibility: Simplera Sync works exclusively with the MiniMed 780G pump running software version 6.23 or newer. This is important – if you have older software, it must be updated before using the sensor. Simplera Sync does NOT work with older pumps or with the Smart MDI system (the Simplera sensor without the “Sync” suffix is intended for inPen injectors).
Wear time: Up to 7 days on a single sensor – the same as Guardian 4.
Measurement frequency: Every 5 minutes, 24/7, without fingersticks (the need for glucose meter calibration has been eliminated in the integrated 780G closed-loop system).
Communication: Bluetooth Low Energy (BLE) – energy-efficient data transmission directly to the pump and the MiniMed Mobile app.
Insertion: A two-step process taking about 9 seconds – the inserter is built-in, no need to purchase it separately.
Patient age: CE certified, indicated for patients from 2 years of age. Medical device class IIb.
Reimbursement (NFZ): The sensor is reimbursed. Patients over 26 years old pay 30% of the price (the monthly reimbursement limit is PLN 510). Children and young people under 26 use separate reimbursement codes.
What is it like in everyday use?
The biggest change compared to Guardian 4 is the elimination of the transmitter. There’s nothing to charge anymore, no shell that can fall off along with the adhesive. The sensor itself is relatively small and has a discreet, contoured housing.
Initial experiences from users and testers indicate that in the first few to several hours after insertion, discrepancies compared to glucose meter readings may occur—especially if there was minimal bleeding during insertion. This is a phenomenon known from many CGM sensors and is worth being prepared for.
It’s also worth knowing that Simplera Sync is typically sold in packs of five. When purchasing smaller quantities from medical stores, the sensor may be removed from the original bulk packaging.
Adhesive patch – is it necessary?
Medtronic does not require the use of an additional patch applied over Simplera Sync. The sensor has its own adhesive base. However, for active individuals or those with sensitive skin, it’s worth having a thin tape or protective patch on hand, especially toward the end of the 7-day wear period when the original adhesive may lose strength.
Simplera vs Simplera Sync – don’t mix up the models!
This is the most common point of confusion when purchasing. There are two different products on the market:
Simplera (MMT-5100) – a sensor designed for the Smart MDI system (works with the InPen smart injector). It does not work with the 780G pump.
Simplera Sync (MMT-5120) – a sensor designed exclusively for the MiniMed 780G pump (software 6.23+). It does not work with InPen injectors.
Before purchasing, always check the product code and make sure you are buying the correct model for your treatment system.
New: Simplera Sync approved by the FDA
In April 2025, Medtronic received FDA approval for the Simplera Sync sensor as part of the MiniMed 780G system for the U.S. market. A limited launch in the U.S. is planned for fall 2025. European users of the 780G pump already have access to this sensor earlier – in Poland it is available for purchase with NFZ reimbursement.
Who is Simplera Sync for – a short summary
Simplera Sync is a good choice if:
You use the MiniMed 780G pump with software version 6.23 (or are ready to update it),
You want to simplify your setup – one component instead of sensor + transmitter,
You value quick and easy insertion without additional accessories,
You benefit from NFZ reimbursement and want a device covered by funding.
Simplera Sync is NOT the right choice if:
You have an older Medtronic pump (e.g., 640G, 770G) – the sensor will not be compatible,
You are treated with multiple daily injections (MDI) and use a pen – in that case, look for the Simplera (MMT-5100) sensor for Smart MDI.